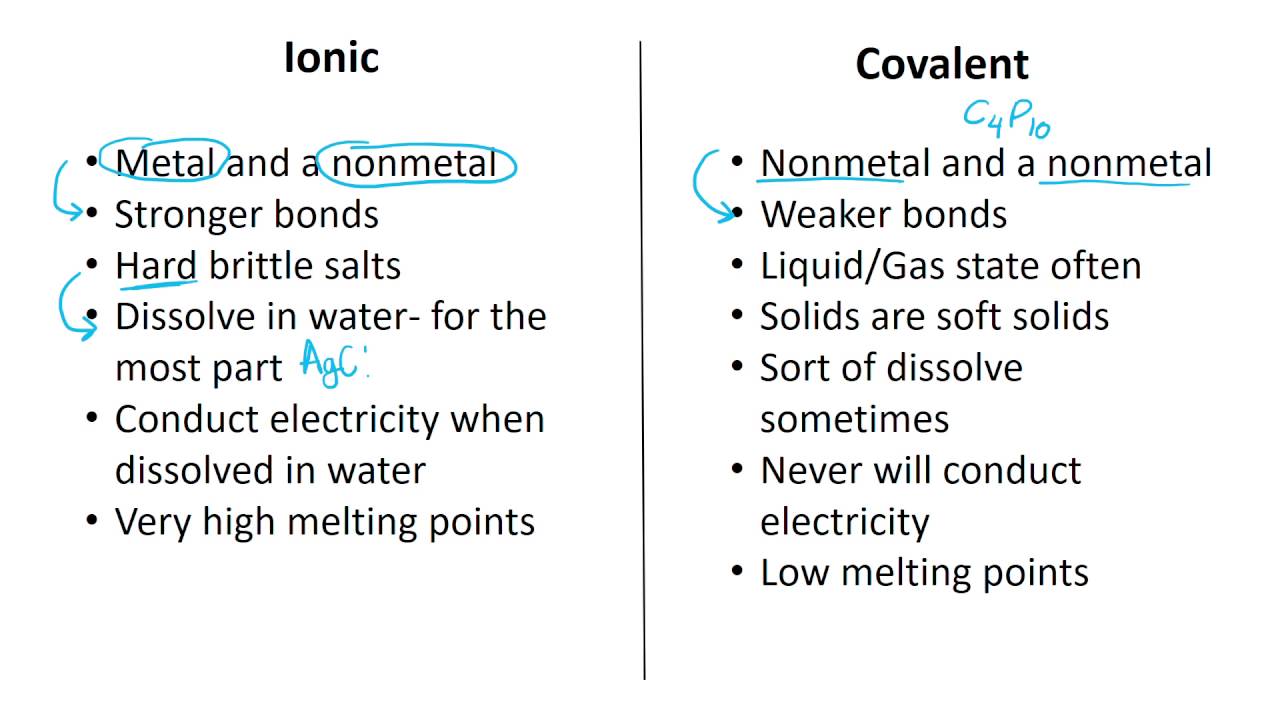
The shared electrons will spend time circulating between the valence shells of the bonded atoms.Ĭovalent bonds form between nonmetals. 2 hydrogen atoms can bond together by sharing their electrons, thus creating a stable valence shell between them with 2 electrons (H2).Ĭovelent bonding leads to the formation of molecules, or formations of atoms that share electrons. 
The motto of atoms that like to form covalent bonds in “sharing is caring!” These atoms resolve the issue of their unfilled outer shells by sharing electrons with other atoms that also need to fill their shells.įor example, hydrogen atoms have 1 electron each, and need 2 for a stable valence shell. Let’s check them out! Covalent Bonds Image: wikipedia There are two common ways atoms like to bond. If this shell does not reach full capacity, it likes to bond with other atoms in order to stabilize. The outermost shell is called the valence shell, and the electrons in this shell are called valence electrons. Each shell can only hold a certain number of electrons, and the shells fill in order from the interior shell outwards. They also have shells that hold the electrons in orbit around the protons and neutrons in the center. Protons have a positive charge, neutrons have a neutral charge, and electrons have a negative charge.Ītoms have an equal amount protons and neutrons so that their charge balances out. But which bond is the best bond? James Bond? That’s silly he’s not an atom at all! Now back to the chemistry! Science!Īll atoms are made up of protons, neutrons, and electrons. If you want to stick a couple of atoms together you would probably use one of two chemical bonding choices – ionic or covalent bonding.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
